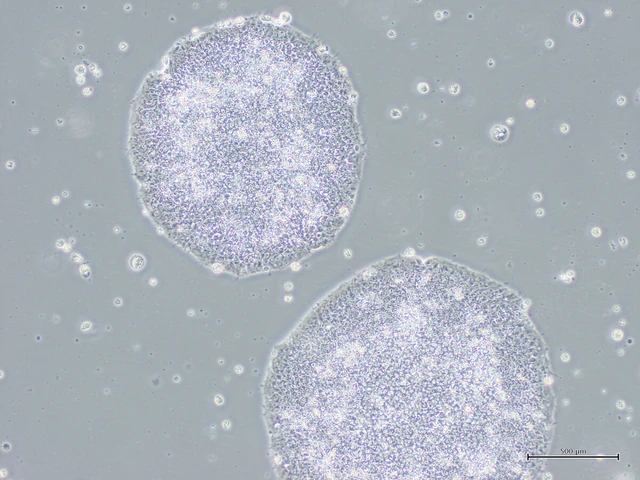
iPS cells cultured by Sumitomo Pharma Co. Photo: Courtesy of Sumitomo Pharma
The world's first permission of iPS cell-derived medicine, albeit provisional, was given to Tokyo-based startup Cuorips Inc.'s cardiac muscle cell sheet called "ReHeart" and Osaka-based drugmaker Sumitomo Pharma Co.'s "Amchepry" dopaminergic nerve cells.
The heart disorder and Parkinson's disease therapies will be covered by the country's public health insurance after the companies file respective applications for the insurance coverage with the ministry.
To obtain full approval, however, Cuorips, a spinout from the University of Osaka, and Sumitomo Pharma have to gather more clinical data including on efficacy over the next seven years and submit them to the ministry for screening, with the target number of ischemic cardiomyopathy patients to be treated with ReHeart set at 75 and that of Parkinson's disease patients to take the Amchepry therapy at 35.
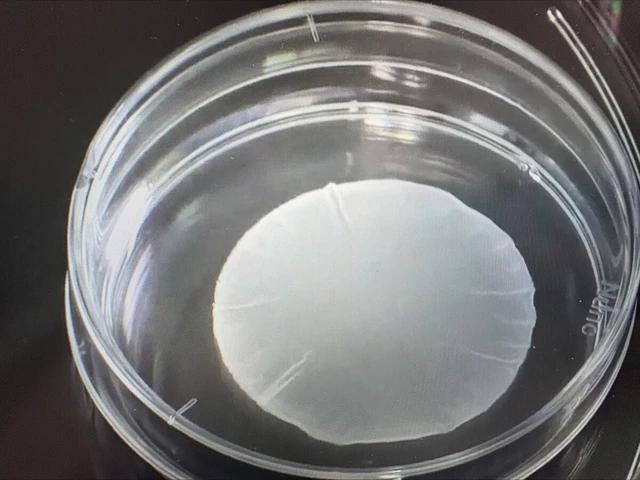
A sheet of heart muscle cells made from iPS cells by University of Osaka startup Cuorips Inc. Photo: Courtesy of Curorips
According to Cuorips, a 4- to 5-centimeter-diameter, 0.1-millimeter-thick ReHeart patch with heart muscle cells made from iPS cells, which can develop into almost all tissues, will restore cardiac function after being applied to a patient's heart.
Sumitomo Pharma said that five million to 10 million Amchepry dopaminergic neural progenitor cells derived from iPS cells are to be transplanted into the center of the brain to improve motor functions.
The ministry's expert panel proposed in February that the ministry give its conditional, time-limited approval to the two therapies ahead of full permission, which takes time.
At a press conference Friday, health minister Kenichiro Ueno said, "I hope patients all over the world are saved."



Max: 1500 characters
There are no comments yet. Be the first to comment.